 These elements have higher electronegativities than Aluminium and Silicon, even though Aluminum and Silicon are above Gallium in Germanium in their respective groups. Gallium and Germanium are exceptions to this rule, though. 
On the other hand, as you progress down a group, the electronegativity decreases, because the number of electron shells increases, counteracting the effect of any increased numbers of protons. This means there is a greater charge which attracts on the shared electrons. In general, electronegativity increases across a period because the number of protons increases while the number of shells stays the same. The elements range in value from 0.7 (caesium and francium), the least electronegative, to 4.0 (fluorine), the most electronegative.Įlectronegativity Patterns and Trends on the Periodic Table He created the Pauling scale, giving each element a value that corresponds to its ability to attract electrons. The greater the number of electron shells, and the further they are away from the nucleus, the less the attractive force will be between the nucleus and the electrons in the covalent bond.Įlectronegativity was first measured and developed by Linus Pauling in 1932. However, this is counteracted by the shielding effect of electron shells. The greater the number the protons, the higher the positive charge of the nucleus and the greater ability the atom has to attract shared electrons. Electronegativity is influenced by the number of protons in the nucleus (atomic number) and the number of valence electrons. We use the term electronegativity to describe the ability of an atom to pull these electrons toward it. In covalent bonds, shared electrons are pulled towards one of the atoms more than the other. 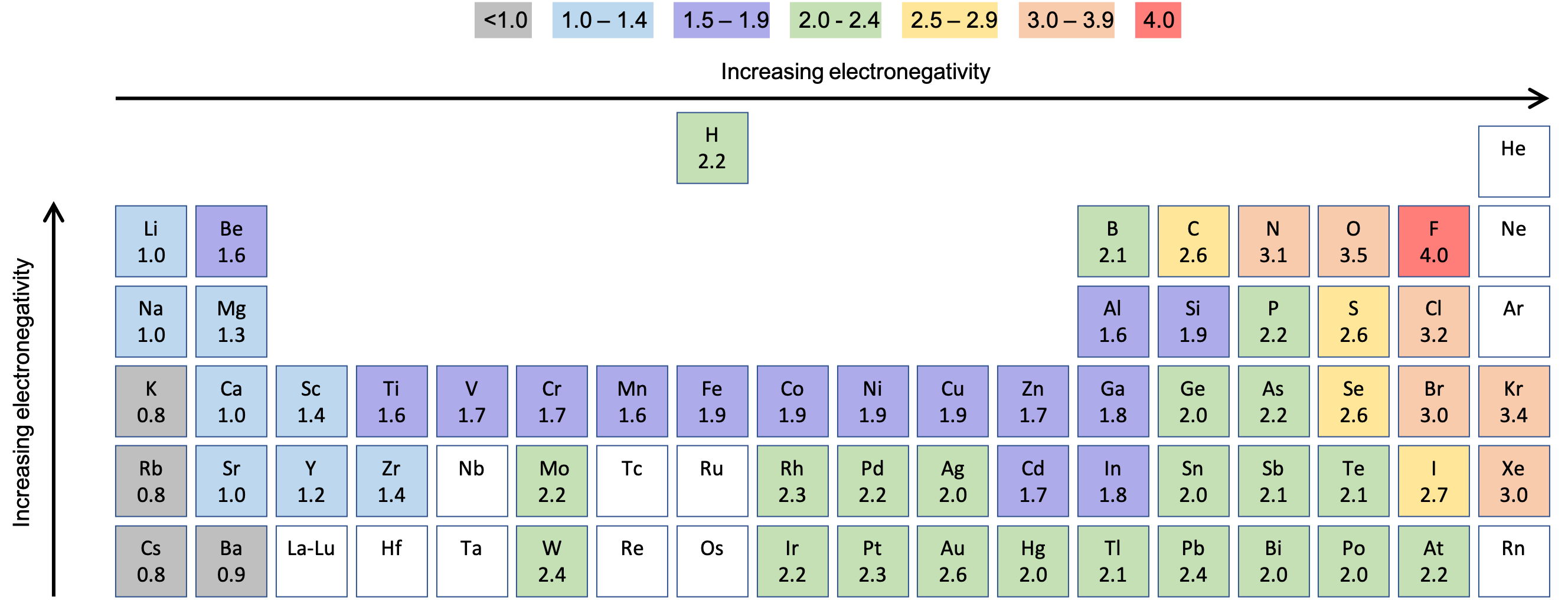
What factors affect electronegativity? Nuclear charge, atomic radius, and shielding (number of electron shells) are three factors that significantly impact the electronegativity value of an element. Which element has the lowest electronegativity? Francium has the lowest electronegativity. Which element has the highest electronegativity? Fluorine has the highest electronegativity. Values between 0.0 and 0.5 are considered nonpolar covalent bonds. Values between 0.5 and 1.6 are deemed polar covalent bonds. Electronegativity difference values greater 2.0 indicate an ionic bond. How do you calculate electronegativity difference? In order to determine the bond type of a compound, you subtract the electronegativities of the bonded elements. 
Covalent bonds involve a pair of electrons, shared between two atoms.įAQs What is electronegativity of an element? Electronegativity is the measure of an atom’s ability to attract electrons towards it in a covalent bond. An ionic bond is a bond between two atoms with opposite charges, where no electrons are shared. Atomic number is the number of protons in the nucleus of an atom. 
Elements in the same group (column) have the same number of outer electrons, while Elements in the same period (row) have the same number of electron shells. Because of this, where an element is on the periodic table can tell you about its chemical properties. Refresher Elements on the periodic table are arranged based on their electronic configurations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
